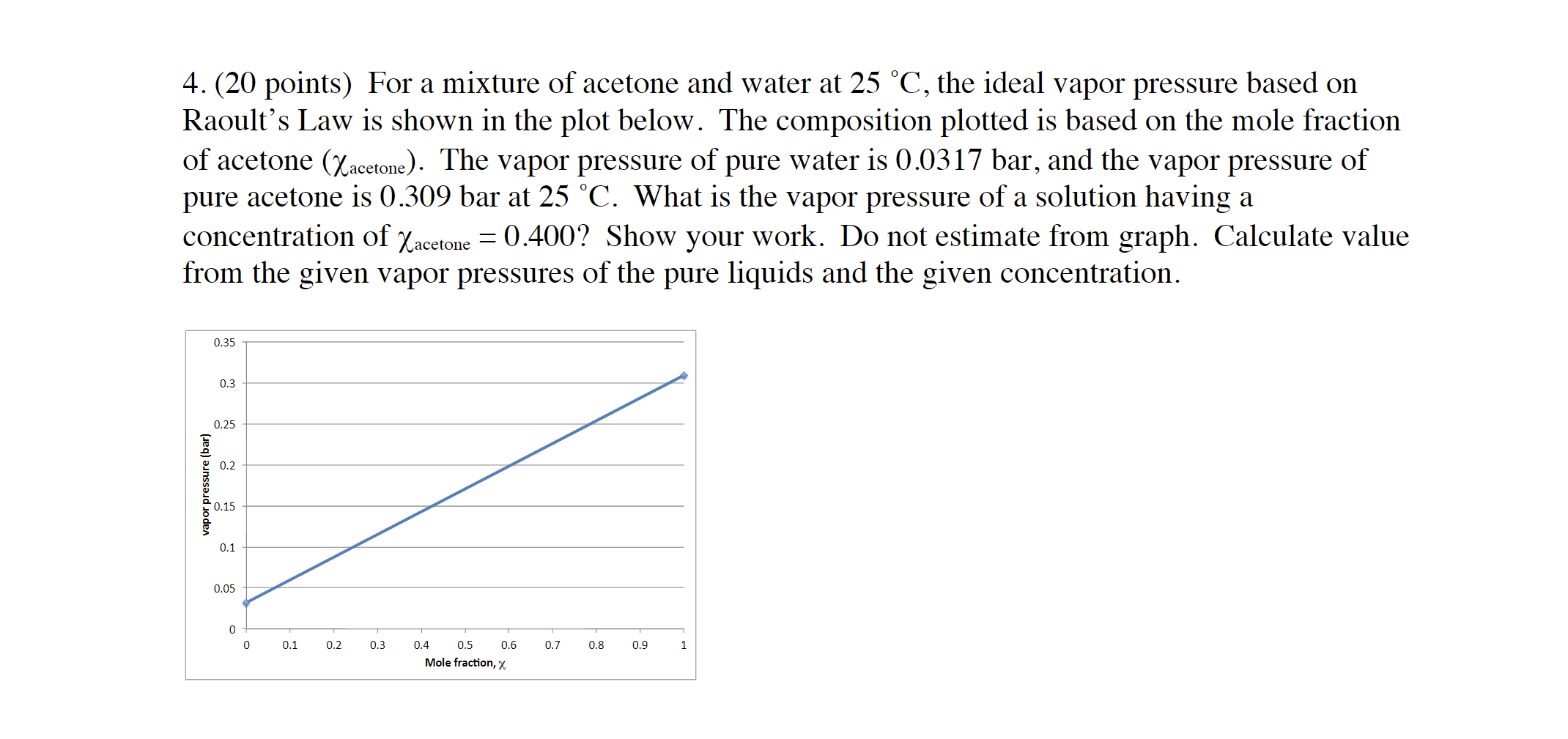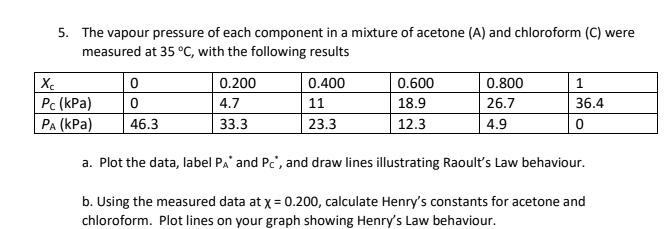
SOLVED: The vapor pressure of each component in a mixture of acetone (A) and chloroform (C) was measured at 35°C, with the following results: 0.200 0.400 0.600 0.800 Pc (kPa) PA (kPa)

Use the following vapor pressure diagram to estimate the partial pressure of acetone. | Homework.Study.com
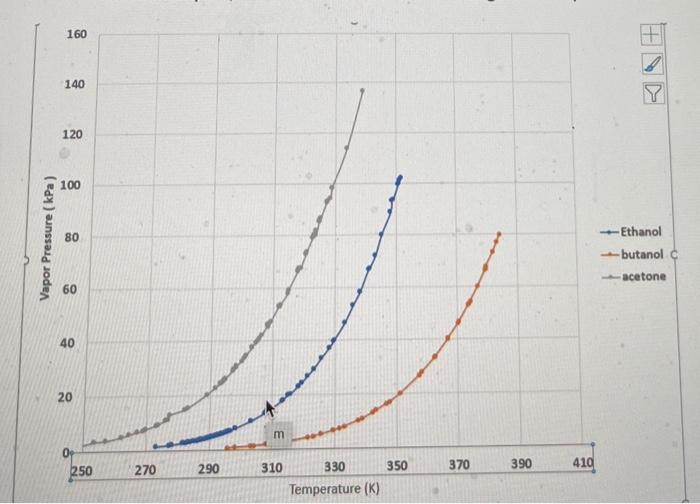
Equilibrium phase diagram of acetone/ethanol mixture under the pressure... | Download Scientific Diagram

SOLVED: At 32°C, the vapor pressures of acetone and toluene are 869 atm and 0.135 atm respectively. Calculate, using Raoult's Law, the mole percentages of acetone and toluene in the vapor for

puaso, Wan cquunon solution of liquids is made. At 30°C, the vapor pressure of pure ether is 646mm and of pure acetone is 283mm. Calculate the mole fraction of each component in

The vapour pressure of acetone at 20^∘ C is 185 torr. When 1.2 g of a non - volatile substance was dissolved in 100 g of acetone at 20^∘ C, its vapour

The vapor pressure of acetone at `20^(@)C` is 185 torr. When `1.2 g` of a non-volatile solute - YouTube

JEE MAINS 7TH JAN 2020 SHIFT 1- At 35°C, the vapour pressure of CS2 is 512 mm Hg and that of aceton - YouTube

SOLVED: Text: For a mixture of Acetone in Water: 1) Calculate the vapor pressures of each component over this mixture over the range between their boiling points. 2) Calculate the liquid and
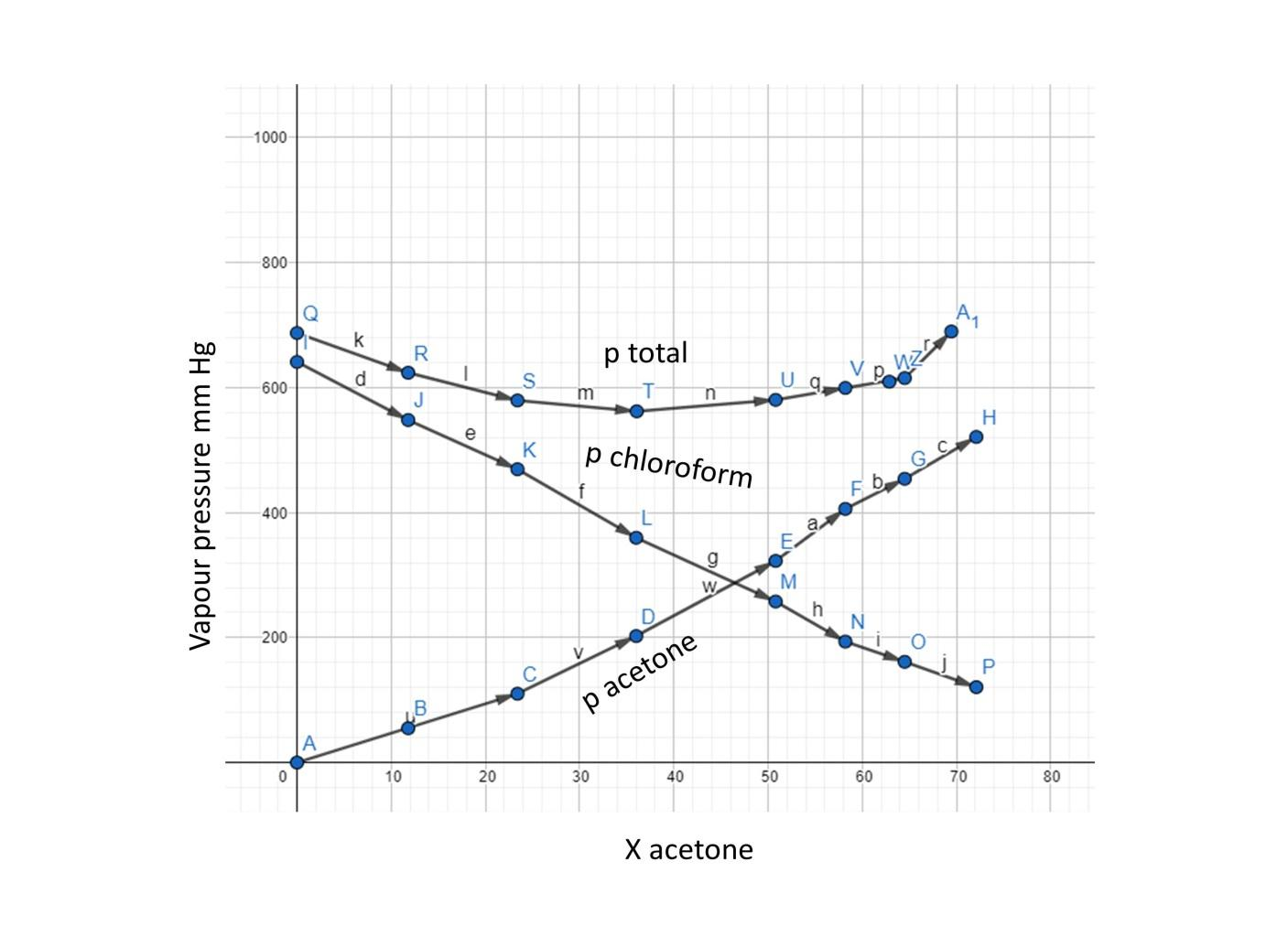
Vapour pressure of pure acetone and chloroform at $328\\,K$ are $741.8mmHg$ and $632.8mmHg$ respectively. Assuming that they form an ideal solution over the entire range of composition. Plot ${P_{total}},{P_{chloroform}}$ and ${P_{acetone}}$ as
The vapour pressure of acetone at 20°C is 185 torr. When 1.2 g of a non-volatile substance was dissolved in 100 g of acetone at 20°C, - Sarthaks eConnect | Largest Online Education Community

QUESTION 2.37 Vapour pressures of pure acetone and chloroform at 328 K are 741.8 mm Hg and 632.8 mm Hg respectively. Assuming that they form ideal solution over the entire range of

The vapour pressure of acetone at \( 20^{\circ} \mathrm{C} \) is 185 torr. When \( 1.2 \mathrm{~... - YouTube